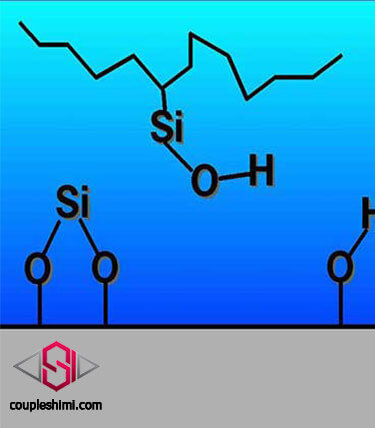
Comparison of COUPSYL® lithium silicate based concrete hardeners with other alkaline silicates
Comparison and differences of lithium silicate with alkali sodium and potassium silicates As you know, lithium silicate based hardeners have high penetration power and after the